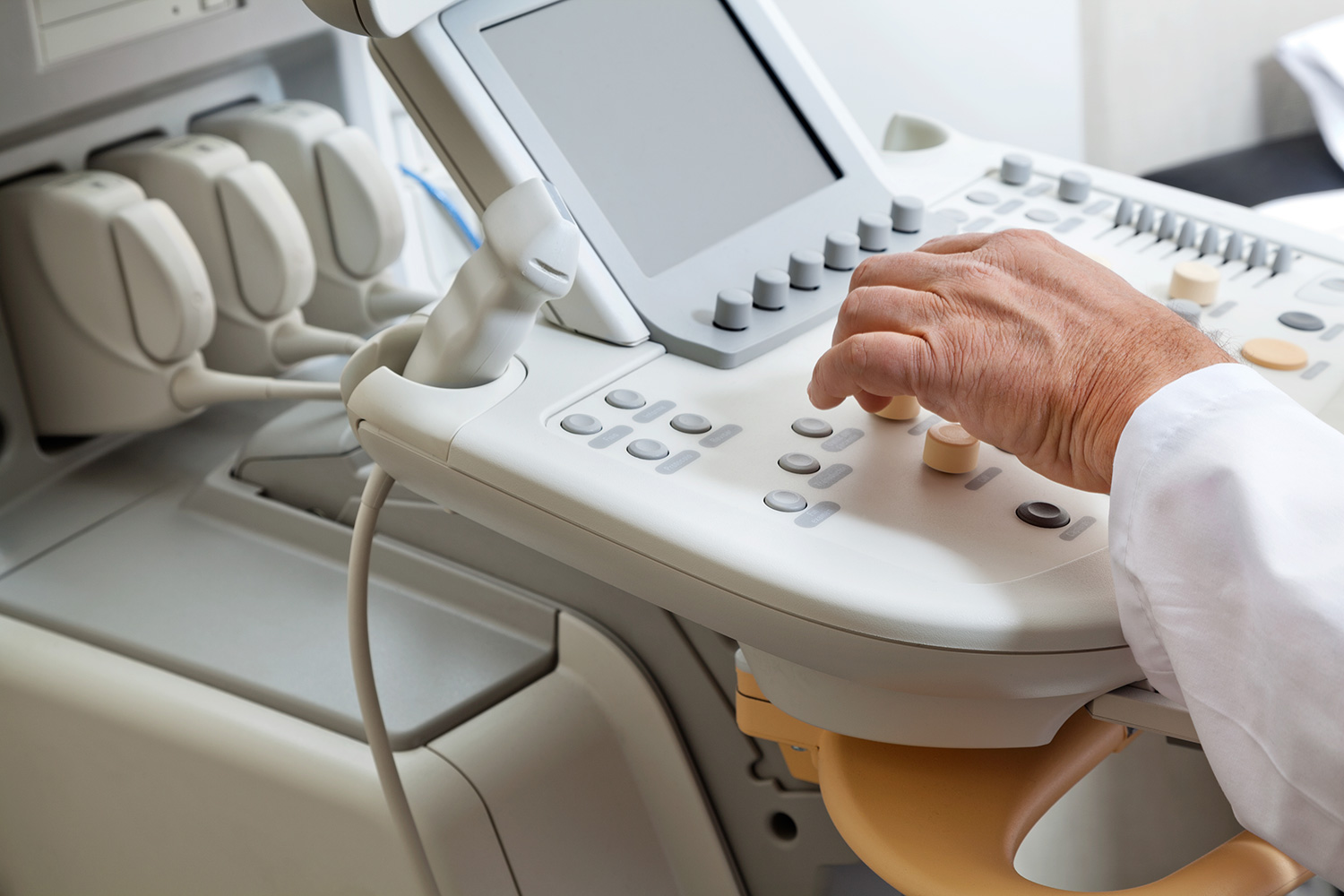
In order to achieve satisfactory measurements, medical devices must be calibrated to identify and correct uncertainties. There is great importance to the accuracy of a device, as it can have a significant impact on test results.
We rely on calibration to ensure accurate measurements, and accurate measurements are critical to the quality, safety, and innovation of most products and services we use and rely on.
Any industry is adversely affected by inaccurate readings and measurements. Nevertheless, the impact can be devastating when it comes to the medical field. Out-of-tolerance medical equipment can produce test results that are either above or below the actual results.
The Need for Calibration
During calibration of medical devices, measurements made by an instrument used in a hospital or other healthcare facility are compared to known standards to determine the accuracy of the measurements.
Physicians need accurate and reliable medical measurements in order to make informed decisions regarding the health and well-being of their patients. Therefore, to maintain confidence in the functioning and operation of the devices used in hospitals, medical centers, and emergency rooms to monitor and treat heart conditions, periodic safety and performance checks are required.
The calibration of medical equipment plays a vital role in ensuring the highest level of measurement accuracy, which is a crucial component of compliance.
- It is imperative to regularly test medical devices for safety and performance, which includes traceable calibration.
Benefits of Regular Calibration
It is beneficial to calibrate medical equipment for several reasons including:
- Increase the effectiveness of the medical instrument.
- Extend the life of the medical instrument.
- Patient and operator safety is maximized.
It is critical to ensure that devices such as electrocardiograms (EKGs), oxygen finger-tip monitors, and defibrillators are regularly tested against national and international standards.
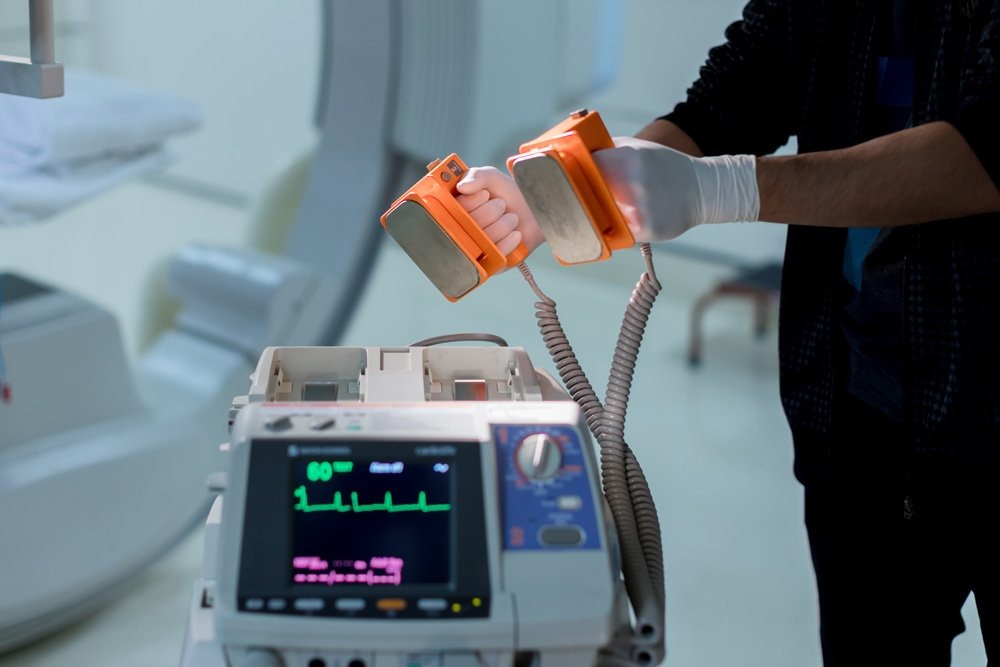
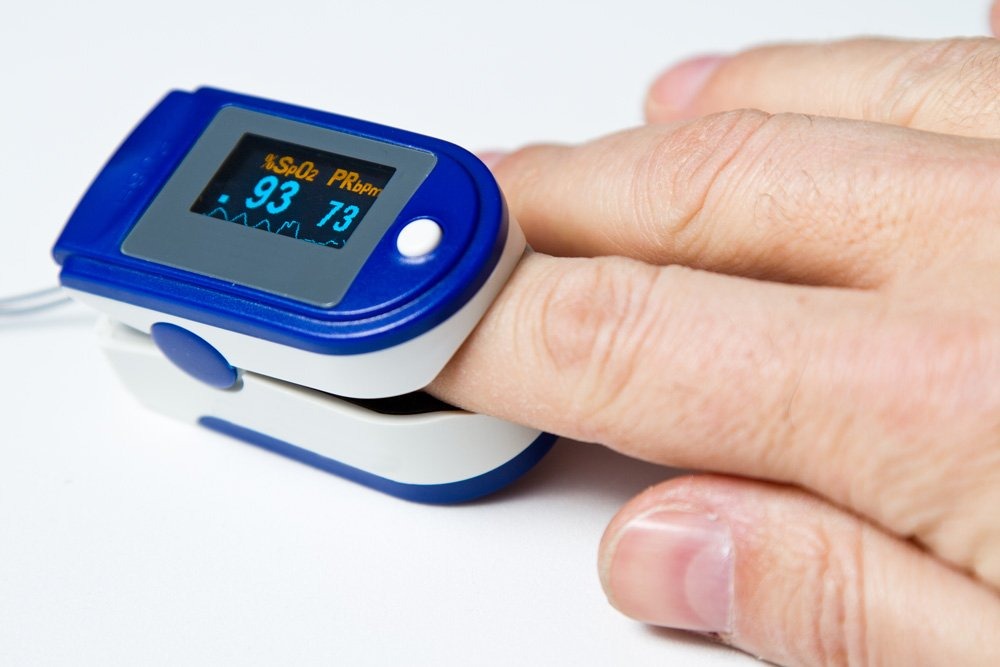
Calibration and Cardiovascular Health
The Center for Disease Control (CDC) estimates that approximately 697,000 Americans die each year from heart disease. According to the World Health Organization (WHO), cardiovascular disease is the world’s number one cause of death, killing over 17 million people every year.
Often considered to be a condition for the older generation, heart disease and other associated illnesses are on the rise across all age groups.
Between 2017 and 2018, the United States spent approximately $229 billion on heart disease.
As a result, hospitals, emergency departments, and health care providers are working to reduce healthcare costs associated with cardiovascular disease without sacrificing quality of care or diagnosis accuracy.
Over time, even the most advanced and expensive medical measuring instruments and equipment may lose their accuracy due to a variety of factors, including everyday wear and tear, environmental changes, and human error.
February is Heart Month
The importance of heart health is something we are proud to highlight. Each year, National Heart Health Month is observed in February. The creation of awareness about the different heart conditions contributes greatly to the reduction in the death rate from heart-related diseases.
Why Choose Allometrics for your Calibration Needs?
In summary, regular calibration of your equipment plays a vital role in providing fast high-quality service along with optimal patient care.
We have highly trained, experienced staff at your disposal to assist you in implementing a calibration schedule. You can have piece-of-mind in knowing that you have seasoned professionals working hard to keep your measuring devices in compliance.





