Hospitals and COVID-19 testing laboratories are working overtime to diagnose and treat patients. The overwhelming amount of new and active cases at some of these facilities puts added pressure on healthcare workers to try and maintain their work environment safe.
The World Health Organization updates its safety guidelines and protocols as new information arises. These guidelines include environmental sampling best practices.
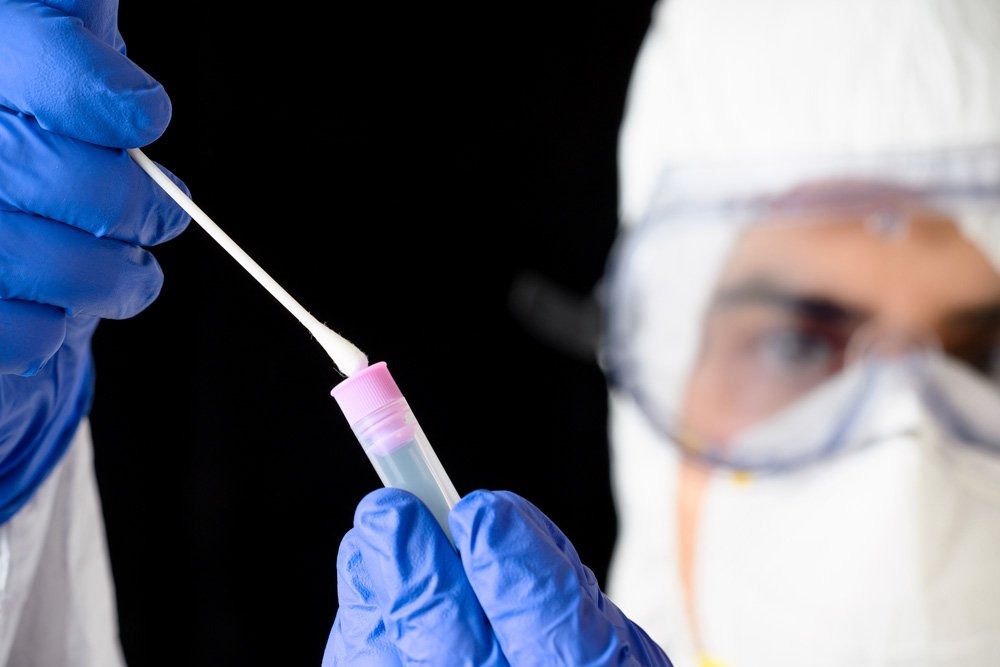
Testing for SARS-CoV-2 is an essential part of the safety protocol of any healthcare facility. Hospitals, clinics, laboratories, and physician offices need to collect samples of surfaces with a certain regularity to prevent the spread of the virus, which causes COVID-19.
Where to Test
Molecular testing for COVID-19 is necessary for any setting in which a patient receives treatment, including hospitals and clinics, as well as households and other private areas such as hotel rooms.
Of the aforementioned, hospitals and public healthcare facilities require a more thorough sampling as the risk of contagion is higher, given that there may be several infected patients at the same time.
The World Health Organization (WHO) recommends the following areas be tested regularly for COVID-19:
Ambulances: Handle and the exterior surface of the medic bag, the inside of blood pressure cuff, the wall next to the patient stretcher, front of the defibrillator, and handlebars.
Hospital Public Areas: Entrances, waiting rooms, and examining rooms require frequent testing. High touch surfaces such as door handle, faucet handle, keyboards, reception, and nurse area desk surfaces, keyboards, light switches, X-ray table. Additionally, ventilation exits, or air purifier filters also need testing.
Patient Rooms: Testing patient rooms is a priority for healthcare facilities, as this is where the highest concentration of the virus could be. A thorough examination of the room will include surface testing of doorknobs, nightstands, bed railings, bed controller, call button, floor, tubing, masks and filters of aerosol-generating procedures, and equipment control panels.
How Frequently Should Hospitals and Labs Test for COVID-19?
The testing frequency depends on many factors, including the number of patients or potential patients treated at the facility.
However, ideally, patient room sampling should take place every day starting from the day of diagnosis and until at least seven days after the release or passing of the patient.
In hospital settings, environment testing should take place before and no more than 24-hours after any aerosol-generating procedure.
In the event of an extensive outbreak, the sampling interval can be increased from every day to every 2-3 days. Additionally, providing the testing is of sufficiently high-quality, hospitals can choose to sample one or two patients over sampling all patients.
Sampling requires using a swab with a synthetic tip and a plastic shaft, and sample areas should not exceed 3.87 in2 (25 cm2).
Allometrics provides high-quality molecular testing for COVID-19. Our testing is done via special Test Tube Swab for Molecular Testing (PCR) for SARS-CoV-2 (COVID-19).
All samples collected by our technicians are correctly labeled and shipped to a third-party specialized laboratory. Results turn arounds range from 24-hours to four business days, depending on the client’s preference.
Allometrics also offers surrogate bacterial and air sampling tests, as well as temperature and humidity mapping, which can significantly help identify and reduce bacterial growth.
Call us today to schedule your COVID-19 molecular testing.






