Every year cardiovascular disease kills more than 650,000 Americans making it the leading cause of death.
Additionally, heart disease costs the United States some $219 billion a year. For this reason, hospitals, ERs, and health care providers are striving to reduce healthcare costs associated with cardiovascular disease without compromising the quality of care and appropriate medical diagnosis.
In the last few decades, we’ve seen considerable advancements in medical technology, which have significantly improved the accuracy of medical diagnosis and treatment of cardiovascular disease.
The Need for Calibration
Medical device calibration involves comparing measurements made by an instrument used by a hospital or other healthcare provider against a known standard to determine the accuracy of its readings.
As healthcare costs continue to rise, and given that people’s lives are at stake, healthcare providers are beginning to understand that the maintenance of their medical devices alone, without calibration, is not enough to ensure proper function and adequate and reliable measurements.
The devices used in medical centers, hospitals, and ERs for monitoring and treating heart conditions require periodic safety and performance checking to have confidence in their functioning and operation.
Accurate and reliable medical measurements are vital for physicians to make appropriate decisions regarding their patients’ health and well-being.
Failure to ensure accurate measurements directly impacts the quality of the care provided to patients. It is likely to increase healthcare costs substantially by possibly subjecting patients to unnecessary medical treatment or, worse yet, misdiagnosing and denying treatment to those who require it.
Regular safety and performance testing that includes traceable calibration in medical devices is a key factor in improving public health. It is likewise a crucial part of compliance as proper calibration ensures the highest level of measurement accuracy.
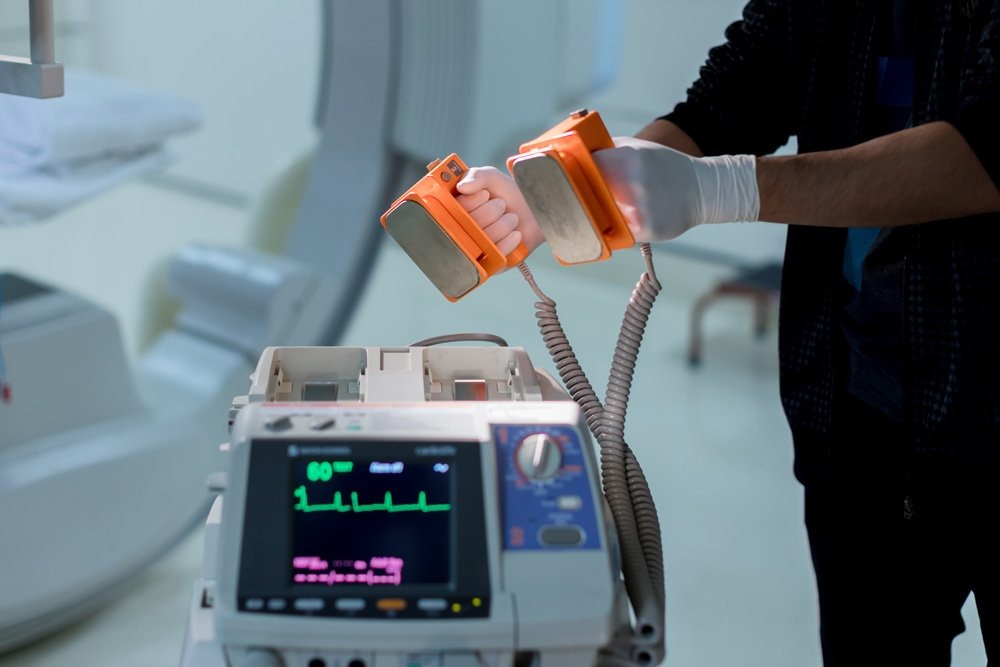
Which Cardiovascular Medical Devices Need Calibration?
Even the most advanced and expensive medical measuring instruments and equipment are affected by the effects of everyday wear and tear, environmental changes, and user error, causing them to lose accuracy over time.
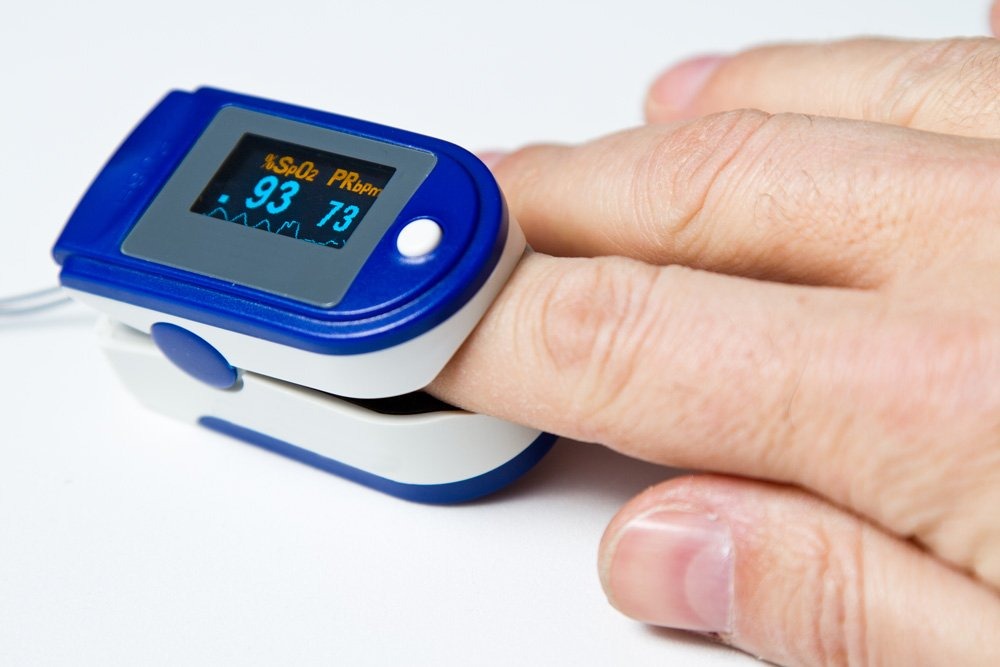
When a patient arrives at a hospital or ER with symptoms compatible with a heart attack, doctors rely on instruments to diagnose, monitor, and revive patients. That is why providing regular testing against national and international standards of devices such as electrocardiograms (EKG), oxygen finger-tip monitors, and defibrillators becomes essential to guaranteeing reliable measurements.
Calibrating medical equipment has many advantages: maximizing the instrument’s effectiveness and extending its lifetime, to minimizing risk for patients and operators.
The Effects of Poor Measurements in The Medical Field
Inaccurate readings and measurements negatively affect any industry. However, when it comes to the medical field, the impact can have dire consequences.
We must remember that physicians use test results to reach a diagnosis. If the readings from the tests are inaccurate, it can lead to an incorrect diagnosis.
Medical equipment that is out of tolerance can shift test results by showing data that is either above or below the actual value. Faulty test results will inevitably alter the diagnosis and treatment course, leading doctors to prescribe higher or lower doses of medication than needed or even prescribe the incorrect medication altogether, potentially causing unnecessary pain, disability, and even patient death.
Medical errors resulting from the use of non-calibrated equipment can then evolve into negligence and malpractice lawsuits.





